Studyfyr
Governed Clinical Research Operations on FHIR and OMOP
Run reproducible clinical analytics in SPE/TRE-style environments with controlled execution and publication pathways.

Why Teams Choose Studyfyr
Why Teams Choose Studyfyr
Most teams can build clinical analytics pipelines. Fewer can operate them safely and consistently at scale.
Common blockers
- Reproducibility breaks across teams and studies
- Traceability is fragmented across tools and notebooks
- Governance and privacy controls arrive too late
- Publishing/export paths become manual and high-risk
What changes with Studyfyr
- Manifest-backed reproducibility by default
- Governed workspaces with controlled lifecycle
- Metadata-only audit patterns (no PHI in audit by design)
- Controlled output and publication workflows in secure environments
How It Works
1.
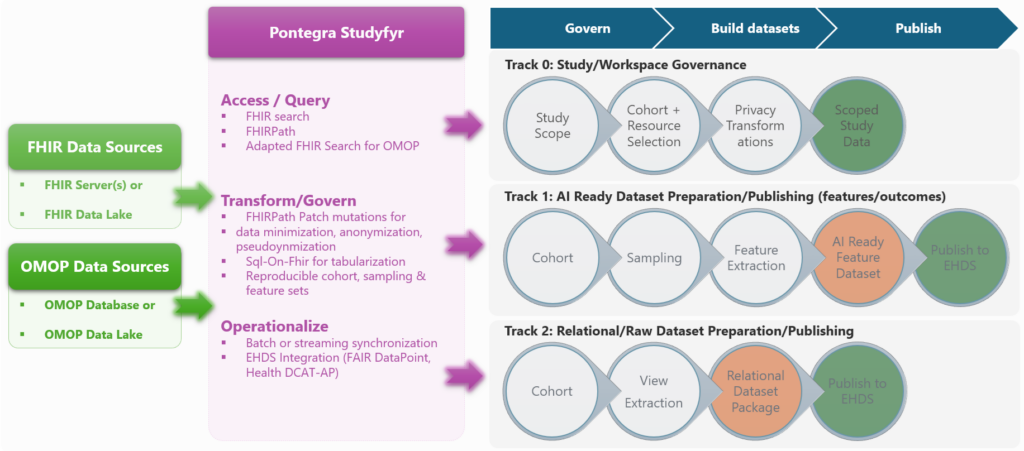
Build with Clinical Research Toolkit (CRT)
Define and execute notebook-friendly pipelines on FHIR and OMOP.
- Connect to FHIR sources – server or lakehouse
- Query like FHIR, compute like Spark
- Transform, minimize, anonymize, pseudonymise resources safely via FHIRPath Patch like mutations
- Extract analysis-ready tables with SQL-On-Fhir like extractions
- Create and execute cohort and dataset workflows
2.
Execute with governance
Run in policy-aware workspaces with managed artifacts (e.g. cohort, dataset), traceability, and explicit refresh/recompute behavior.
3.
Review and publish
Route outputs through controlled review and approved publication/egress workflows
Core Capabilities
Processing
- Native pipeline support for FHIR and OMOP
- Cohort/dataset workflows for analytics and ML preparation
- Large scale, distributed execution via Apache Spark with reproducible run metadata
Governance
- Study-scoped workspaces for specs, runs, artifacts, and audit
- Metadata-only audit defaults (no PHI in audit by design)
- Controlled output and publication patterns for regulated environments



Where This Solution Fits Best
Who is it for?
Hospitals and academic TREs
National/regional secondary-use programs
Life sciences RWE and observational analytics teams
Population health programs with governance requirements
Frequently asked questions
Is Studyfyr replacing CRT?
No. CRT is the open-source analytics toolkit. Studyfyr is the governed platform layer for enterprise operation.
Can we run Studyfyr in our own environment?
Yes. Studyfyr is designed for controlled customer environments where data remains in place.
Does Studyfyr support pseudonymized research workspaces?
Yes. Studyfyr supports policy-driven privacy transformations during governed workflows so researchers can work in notebooks and submit airlock requests in the same controlled workspace.
How are governance and lineage handled?
Studyfyr uses manifest-backed run history, managed artifact lifecycle, and controlled workflow patterns so execution and outputs are traceable.
Is Studyfyr officially EHDS certified?
Studyfyr is positioned as an EHDS-aligned implementation platform for secondary-use operating patterns. Final compliance responsibility remains with the implementing organization and applicable national/EU governance processes.
If your organization needs secure, governed, and reproducible clinical research operations, Studyfyr provides a practical operating model from pilot to multi-study delivery.