From EHDS requirements to runnable secondary-use pipelines — secure, auditable, and Spark-scalable.

WHAT PONTEGRA PROVIDES
Governed FHIR and OMOP processing with manifest-backed traceability and controlled output pathways
Pontegra helps organizations implement EHDS-aligned secondary use workflows:
- Secure analysis inside a SPE/TRE
- Auditable access and processing with run manifests and lineage
- Controlled outputs
All while enabling cohort building and dataset/feature extraction at scale and publishing dataset metadata to catalogs within EHDS networks.
Powered by: Studyfyr – The Governed Clinical Research Toolkit and Platform on FHIR and OMOP.
Case Study: This impact is proven at the University of Cologne, where we industrialized laboratory interoperability by scaling from a pilot to a production environment processing millions of laboratory records using Ignifyr. [Learn more]
why thıs matters
Unifying Governed Processing and Publication
Secondary use programs are under pressure to deliver research value while maintaining governance, privacy, and auditability.
Common delivery gaps
- Permit and policy requirements are not connected to day-to-day analytics execution
- EHDS permits are documents, but execution needs operational controls
- Secure processing controls and output controls are handled as separate systems
- EHDS requires audit trails, but notebooks don’t generate manifests
- Reproducibility and traceability are fragmented across tools and teams
- EHDS requires airlock, but most TREs bolt this on late
This results in
12-18 month timelines, compliance incidents, researcher frustration.
Pontegra addresses this with a unified operating model for governed processing and controlled publication workflows.


Where Pontegra Fits in the Operating Model
Roles / Operating Model
Health Data Access Body (HDAB)
- Evaluates secondary-use requests and issues permit/authorization decisions.
- Defines approval conditions (purpose, allowed users, retention, policy constraints).
Health Data Holder
- Provides access to approved data scope under organizational/legal controls.
- Ensures provisioned study data matches authorized purpose and minimization rules.
Data User (Research Team)
- Submits study request with purpose, population intent, and data requirements.
- Executes approved analytics only within controlled workspace boundaries.
SPE/TRE Platform Operator (Studyfyr-enabled)
- Binds request/approval metadata to executable controls in the runtime.
- Enforces policy-aware pipelines, audit/manifest traceability, and controlled egress.
- Promotes only approved outputs as immutable published artifacts.
Governance flow
Governance decisions are converted into machine-actionable study controls, so policy intent is preserved from request to execution to publication.
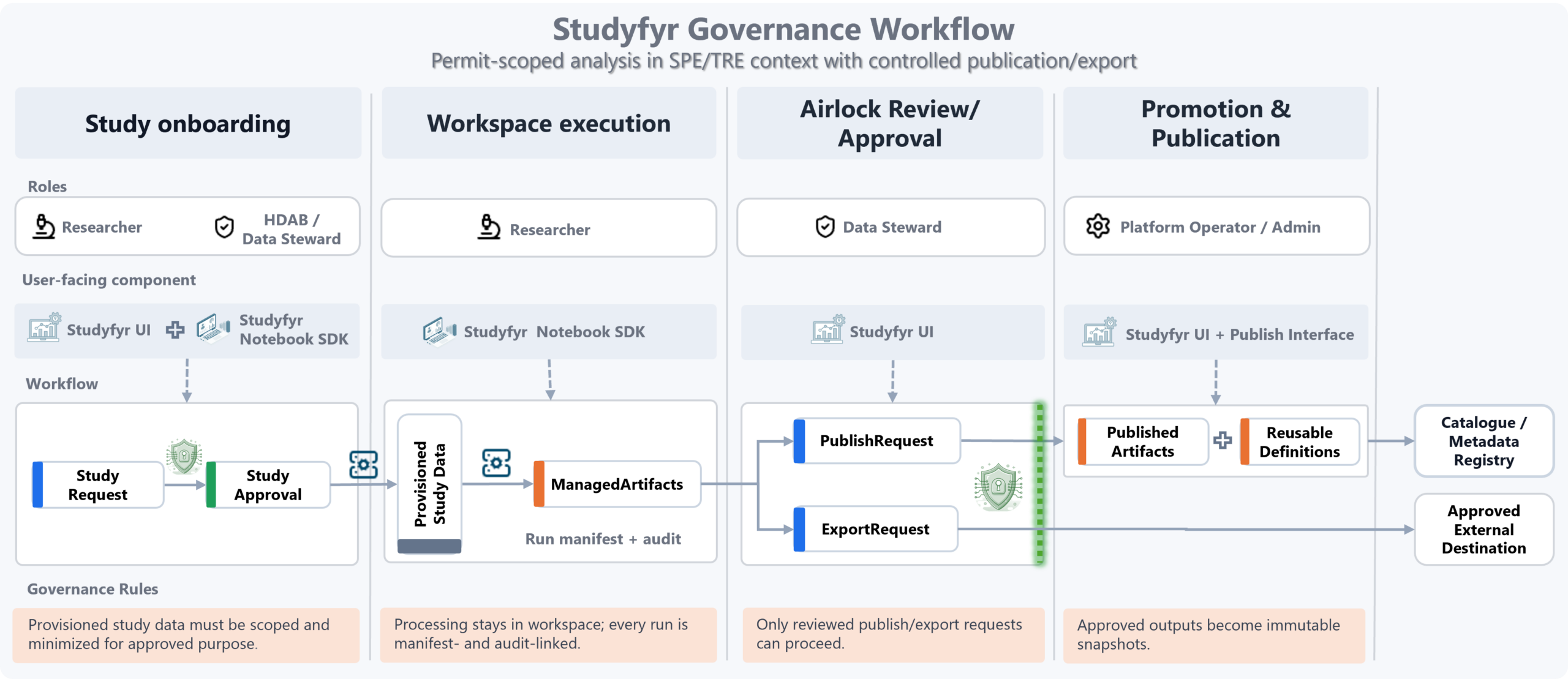
- Onboarding: request and approval define purpose/scope.
- Execution: processing stays in workspace with manifest/audit traceability.
- Airlock: publish/export requires steward review and approval.
- Promotion: approved outputs become immutable published artifacts.
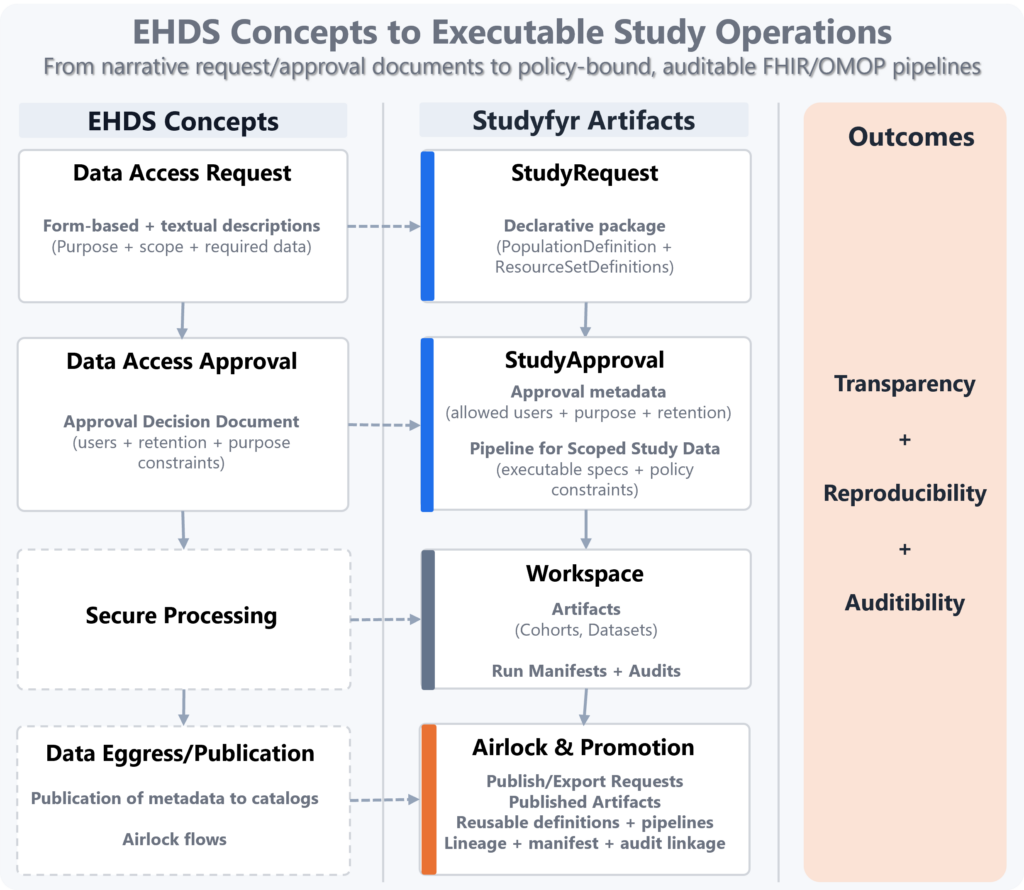
From EHDS Documents to Executable, Auditable Study Operations
Studyfyr binds EHDS governance concepts to executable data operations on FHIR/OMOP, so approvals are not only documents but machine-actionable controls. Result:
- Transparency: explicit request/approval-to-pipeline mapping.
- Reproducibility: the same approved definitions execute deterministically.
- Auditability: manifests and metadata audit events link decision, execution, and published outputs.
EHDS Alignment Checkpoints
| EHDS expectation | Pontegra implementation pattern | Typical evidence artifact |
|---|---|---|
| Permit-gated access and purpose limitation | Study-scoped workflows with explicit approval context | Run manifest with study/run metadata |
| Processing in secure environments (SPE/TRE model) | Controlled in-environment execution near data | Workspace/run records |
| Auditable access and activity | Metadata-first audit events plus immutable run history | Audit event stream + run manifests |
| Controlled outputs and egress | Airlock-compatible review and approval gates | Review decision records |
Note: EHDS-aligned implementation approach; not a blanket legal certification claim
Platform Capabilities
Secure Processing and Auditability
- Governed study workspaces with explicit lifecycle boundaries.
- Policy-driven minimization, anonymization, and pseudonymization controls.
- SPE/TRE-ready Spark execution on study-scoped data under environment controls.
- Manifest-backed run history and metadata audit events for traceability.
Controlled Outputs and Airlock Workflows
- Workspace artifacts (for example, cohorts and datasets) remain inside controlled storage by default.
- Publish/export actions require explicit review and approval (airlock-compatible flow).
- Approved outputs are promoted as immutable snapshots with lineage and audit linkage.
FHIR- and OMOP-Based Analytics at Scale
- Reproducible Spark processing foundations for secondary-use analytics.
- Data access from FHIR APIs, OMOP databases, and FHIR/OMOP lakehouse storage
- FHIR search/FHIRPath filtering and SQL-oriented extraction to analysis-ready tables.
- Unified processing model across FHIR and OMOP sources.
Research Workflow: Cohorts -> Sampling -> Features
- Repeatable cohort extraction with entry/exit/eligibility logic.
- Sampling strategies for periodic and event-aligned timepoints.
- Relational dataset preparation as reusable, well-defined tables/views.
- Feature dataset generation for statistics and ML, including longitudinal/windowed features.
- Deterministic run lifecycle supporting reproducibility and accountability.



WHERE THIS SOLUTION FITS BEST
Where This Solution Fits Best
National/regional secondary-use programs implementing EHDS-style governance
Hospitals and academic organizations operating SPE/TRE environments
Health data holders and platform teams needing permit-bound, auditable analytics
Research and RWE teams requiring controlled cohort-to-dataset workflows on FHIR/OMOP
OUR WORK IN ACTION
Featured Case Studies
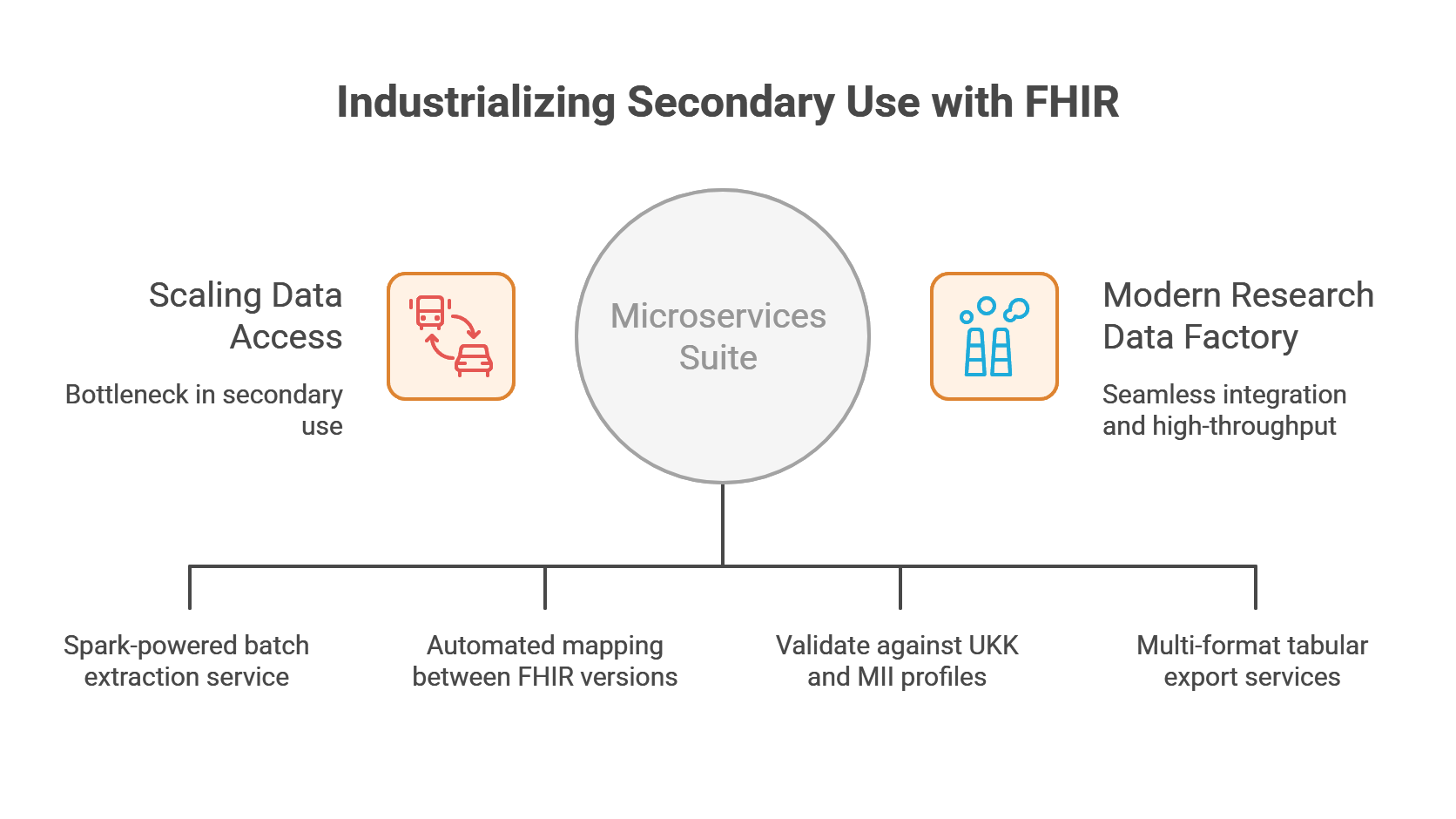
University of Cologne
Industrializing Secondary Use with Advanced FHIR Validation and Export Services
SEE YOUR OPTIONS
Packages
Available now
Design Partner Track
- Best for: Organizations co-shaping early implementation priorities.
- Timeline: Ongoing
- Typical output: Use-case backlog, phased rollout plan, early access alignment.
Planned June 2026 *
Secondary Use Starter
- Best for: Teams ready to implement a first governed cohort-to-dataset pipeline.
- Timeline: 4-6 weeks
- Typical scope: One cohort definition, one dataset specification, pipeline implementation, run manifest/audit outputs, handover.
Planned October 2026 *
Secondary Use Scale-Up
- Best for: Multi-study rollout and recurring governed operations with Studyfyr.
- Timeline: 3-6 months
- Typical scope: Additional cohorts/datasets, scheduled refresh/recompute cycles, operating model hardening.
* Planned dates may be adjusted.